Effective Early Treatments for Managing Hidradenitis Suppurativa Pain and Scarring
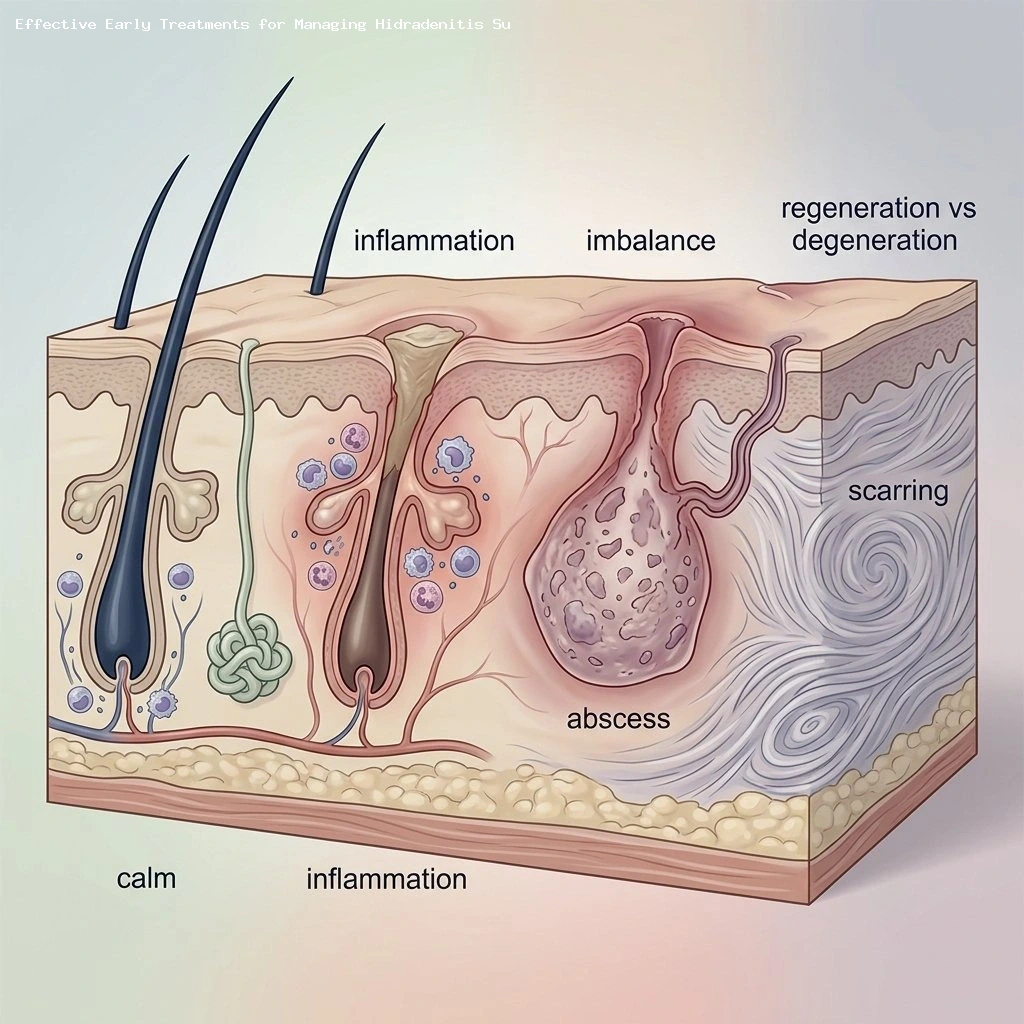
Hidradenitis suppurativa: a disease that’s more than skin deep
Hidradenitis suppurativa (HS) is a chronic inflammatory skin condition that often causes deep, painful nodules, recurrent abscesses, draining tunnels (sinus tracts), and progressive scarring in skin folds such as the armpits, groin, and under the breasts.
The visible lesions are only part of the picture: many people with HS live with persistent pain, ongoing drainage, anxiety about flare-ups, and major impacts on daily life, relationships, and work productivity (Source: American Academy of Dermatology, Hidradenitis Suppurativa Guidelines).
What clinicians discussed at recent case-based roundtables
At three recent case-based roundtable gatherings, three dermatologists — F. George Hougeir, MD; Afsaneh Alavi, MD; and Harrison Nguyen, MD, MBA, MPH — led detailed discussions with dermatology clinicians about challenging HS cases and the role of biologic therapies in routine practice.
The moderators bring different clinical perspectives: Dr. Hougeir practices general dermatology and Mohs surgery in Georgia; Dr. Alavi directs a medical dermatology fellowship at a major academic center; and Dr. Nguyen combines clinical care, Mohs surgery, and research leadership in Texas. Their discussions focused on practical decision-making rather than textbook algorithms.
Why counting lesions isn’t enough
One persistent theme was that simple lesion counts or static categories don’t capture how HS affects a person’s life.
Traditional staging systems such as the Hurley classification provide a basic framework but are limited because they describe structural disease stages rather than current inflammatory activity or symptom burden (Source: American Academy of Dermatology, Hidradenitis Suppurativa Guidelines).
Clinicians at the roundtables emphasized using more dynamic measures — including inflammatory lesion counts, patient-reported symptoms like pain and drainage, and validated scoring tools — to guide treatment choices and track response over time (Source: International Hidradenitis Suppurativa Severity Score System, IHS4 publications).
Tools that help measure real-world disease activity
Two practical measures were highlighted repeatedly:
- HiSCR (Hidradenitis Suppurativa Clinical Response), a common clinical trial endpoint that tracks reduction in inflammatory lesion counts and is helpful when monitoring response to systemic therapies (Source: AbbVie, PIONEER I and II trials).
- IHS4 (International Hidradenitis Suppurativa Severity Score System), a scoring system designed to reflect active inflammatory burden and guide clinical staging in practice (Source: IHS4 development study).
These dynamic tools help clinicians move beyond a single “stage” label and make treatment decisions that reflect present inflammation, pain, and quality-of-life impact.
Biologics are changing the treatment conversation
Antibiotics, hormonal treatments, and procedures (incision and drainage, deroofing, excisions) still have important roles in HS care, but many patients cycle through these options without sustained control.
That pattern can lead to delayed disease control, repeated tissue damage, and cumulative scarring. As a result, the expanding range of biologic therapies has shifted clinician focus toward long-term disease modification rather than episodic symptom suppression (Source: American Academy of Dermatology, Hidradenitis Suppurativa Guidelines).
When to consider biologics: timing matters
A recurring discussion point was timing — specifically, whether earlier biologic intervention can change the course of disease for some patients.
Roundtable attendees noted a “window of opportunity” when early inflammatory lesions or new painful nodules appear, before extensive tunneling or scarring develop. Intervening during that window may preserve tissue integrity and reduce long-term morbidity (Source: Expert consensus from roundtable discussions; clinical rationale reflected in HS guidelines).
Dr. Hougeir and others emphasized that waiting for multiple flares or clear irreversible damage can limit the benefits that systemic therapies offer. Several clinicians said they are increasingly willing to consider biologics earlier for patients with rapid progression, significant pain, or early scarring shown on exam (Source: Expert consensus from roundtable discussions).
Clinical signs that prompt escalation
Factors that commonly triggered consideration of biologic therapy included:
- Recurrent flares despite adequate topical or antibiotic courses.
- Early formation of tunnels or sinus tracts visible on exam.
- Significant, persistent pain or continuous drainage that impairs daily activities.
- Rapid progression of lesions or objective increases in inflammatory lesion counts.
Attendees stressed that the decision to escalate should be individualized, balancing disease activity, previous treatment response, and patient preferences (Source: American Academy of Dermatology, Hidradenitis Suppurativa Guidelines).
Where bimekizumab may fit in treatment pathways
One biologic that generated notable discussion was bimekizumab, an agent that inhibits both IL‑17A and IL‑17F, two inflammatory cytokines involved in HS pathophysiology.
Clinical trial programs for bimekizumab in HS (Phase 3) have shown encouraging HiSCR responses, prompting clinicians to consider it for patients with a high inflammatory burden, frequent flares, or inadequate response to other systemic therapies (Source: UCB, BE HEARD Phase 3 trials press release).
Roundtable participants described thinking about bimekizumab particularly when inflammation — rather than only mechanical issues — is the dominant driver of disease activity. Practical aspects such as the dosing schedule, mode of administration, monitoring for side effects, and upfront patient education were all part of the conversation (Source: UCB, BE HEARD Phase 3 trials press release).
The moderators emphasized that while trial data are promising, long-term real-world experience and head-to-head comparisons will further clarify where bimekizumab belongs in routine practice (Source: UCB, BE HEARD Phase 3 trials press release).
Practical considerations when starting a biologic
Clinicians agreed on several practical steps to improve outcomes when initiating any biologic:
- Discuss realistic expectations about timing of response and potential adverse effects with the patient before starting therapy.
- Use validated measures such as HiSCR or IHS4 to document baseline disease activity and track response over time (Source: AbbVie, PIONEER trials; IHS4 development study).
- Coordinate care for comorbid conditions (e.g., metabolic syndrome, mental health support) that commonly accompany HS and affect outcomes (Source: American Academy of Dermatology, Hidradenitis Suppurativa Guidelines).
- Plan for ongoing monitoring and contingency strategies if response is incomplete or adverse effects emerge.
Real-world experience and the need for individualized decisions
Attendees stressed that no single biologic is right for every patient. Treatment choice should weigh:
- Disease characteristics (inflammatory versus fibrotic features).
- Prior therapy responses and tolerability.
- Comorbidities and safety concerns.
- Patient preferences, lifestyle, and access considerations.
Shared decision-making, clear patient education, and close follow-up were highlighted as essential components of long-term HS management (Source: American Academy of Dermatology, Hidradenitis Suppurativa Guidelines).
Takeaway: timely, sustained, and patient-centered care
The roundtable discussions reinforced several practical messages for clinicians managing HS in real-world settings.
First, static staging alone is not enough. Clinicians should use dynamic assessments — including inflammatory lesion counts and patient-reported symptoms — to guide escalation to systemic biologic therapy when appropriate (Source: IHS4 development study; AbbVie, PIONEER trials).
Second, earlier intervention with biologics may change disease trajectory for selected patients, especially those with rapid progression, significant pain, or early scarring, but timing must be individualized and discussed openly with patients (Source: Expert consensus from roundtable discussions; American Academy of Dermatology, Hidradenitis Suppurativa Guidelines).
Finally, the arrival of newer agents such as bimekizumab expands options for modulating the inflammatory drivers of HS. Ongoing accumulation of long-term and real-world data will refine how clinicians sequence therapies to optimize durability of response and quality of life (Source: UCB, BE HEARD Phase 3 trials press release).
Sources
- American Academy of Dermatology, Hidradenitis Suppurativa Guidelines (clinical guidance and treatment recommendations).
- IHS4 development and validation publications (International Hidradenitis Suppurativa Severity Score System).
- AbbVie, PIONEER I and PIONEER II trials — HiSCR endpoint and pivotal clinical trial data for biologic therapy in HS (press releases and trial summaries).
- UCB Pharma, BE HEARD Phase 3 trials — bimekizumab efficacy and safety results (press release and clinical study reports).
- Expert roundtable summaries and clinician consensus statements from recent HS case-based discussions (moderator and attendee insights).
