Cutting-Edge Treatments Transform Atopic Dermatitis Care in 2026
A new era: targeted medicines are changing how we treat atopic dermatitis
Over the past few years, treatment for atopic dermatitis (AD) has moved from a toolbox dominated by broad anti-inflammatory drugs to one that includes highly targeted options—biologic monoclonal antibodies, oral Janus kinase (JAK) inhibitors, and novel nonsteroidal topical agents.
These newer options are not just additional choices; they have reshaped real-world decision making in clinics. Clinicians now consider earlier use of targeted agents instead of reserving them as last-line treatments, because many of these drugs offer sustained disease control with mechanisms aimed at the specific biology driving a patient’s eczema (Source: American Academy of Dermatology, Atopic Dermatitis Guidelines).
How specialists are thinking about treatment sequencing
Leading clinicians at a recent specialty conference described a clear shift in practice: targeted therapies are increasingly the backbone of long-term management, while traditional agents—like topical corticosteroids and topical calcineurin inhibitors—are often used as short-term bridges or for focal flares.
That shift reflects growing confidence in the safety profiles and durable responses of targeted medicines, and a desire to avoid prolonged, nonspecific immunosuppression when possible (Source: American Academy of Dermatology, Atopic Dermatitis Guidelines).
What “targeted” means in everyday care
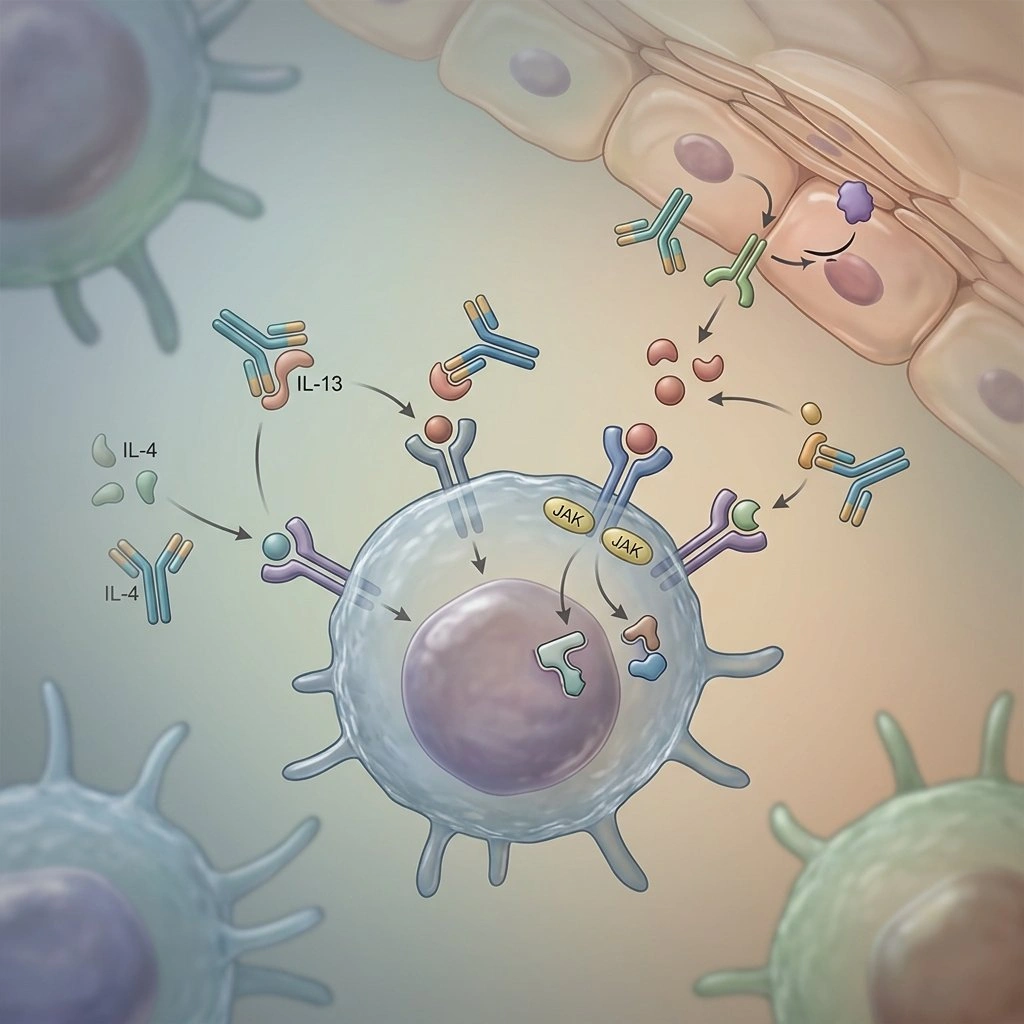
When dermatologists say “targeted,” they mean therapies that interrupt a defined immune pathway known to drive AD symptoms—so instead of broadly dampening immune activity, these drugs block specific cytokines or signaling molecules. This can translate into better itch control, improved sleep, and clearer skin for many patients.
Examples that have altered practice include the anti-IL-4/IL-13 monoclonal antibody dupilumab, and oral JAK inhibitors such as abrocitinib and upadacitinib, all of which have become common options for moderate-to-severe disease (Source: Regeneron Pharmaceuticals; Pfizer; AbbVie press releases).
An expanding therapeutic landscape: what’s available now
The range of approved and investigational options has broadened rapidly, offering clinicians and patients more ways to match treatment to individual needs.
Topical, nonsteroidal medicines—such as tapinarof, topical ruxolitinib, and roflumilast—have given clinicians alternatives for long-term topical control, particularly when minimizing steroid exposure is desired (Source: Dermavant/Incyte/Arcutis press releases).
On the systemic side, oral JAK inhibitors (for example, abrocitinib and upadacitinib) and several biologic monoclonal antibodies (including dupilumab, tralokinumab, lebrikizumab, and nemolizumab) provide highly targeted options for patients with widespread, refractory, or severe disease (Source: Pfizer; AbbVie; Regeneron; LEO Pharma; Eli Lilly; company press releases).
From too few options to too many choices
While having more treatments is good news, it has shifted the clinical challenge: physicians now face complex decisions about which therapy to start, when to switch, and how to sequence treatments for the best long-term outcome.
That complexity calls for careful evaluation of disease features, safety profiles, and patient goals rather than a one-size-fits-all approach.
Personalizing treatment: more than skin deep
Clinicians emphasize that treatment selection is an individualized process. No single factor determines the ideal therapy: disease severity, comorbid conditions (like asthma or allergic rhinitis), prior treatment responses, safety concerns, and patient preferences all matter.
Because atopic dermatitis is a multifaceted disease—affecting sleep, mood, social functioning, and more—visual inspection alone doesn’t capture the whole picture. Listening to patients about itch severity, impact on daily life, and their tolerance for certain risks is essential to choosing the most appropriate option.
In practice, that means many practitioners use a shared decision-making model: clinicians present the reasonable options, explain benefits and risks, and then partner with the patient to choose a path forward.
Systemic inflammation and comorbidity awareness
Modern views of AD increasingly recognize it as a systemic inflammatory disorder rather than a purely skin-limited condition. This perspective helps explain why people with AD often have related atopic or inflammatory conditions.
Because some targeted therapies affect immune pathways common to multiple diseases, their use may influence, positively or negatively, coexisting conditions. As a result, clinicians are factoring comorbidity profiles into treatment choices more often than before (Source: American Academy of Dermatology, Atopic Dermatitis Guidelines).
Experts also note a need for clinical trials that prospectively assess how treatments perform in patients with common comorbidities, to better mirror real-world practice and guide clinicians in selecting therapies that improve overall health, not just skin symptoms.
“We need trials that explicitly include comorbidity assessments so we can begin to parse therapies according to broader patient needs,” one specialist observed.
Spotlight on chronic hand eczema and pediatric care
Two areas getting more attention are chronic hand eczema (CHE) and pediatric atopic dermatitis, both historically challenging to treat and sometimes underrecognized in adult clinics.
CHE is often disabling because of pain, fissuring, and interference with work or caregiving. Recently approved topical agents specifically studied for hand disease—most notably the approval of topical delgocitinib for chronic hand eczema—give clinicians new, evidence-based options for this site (Source: LEO Pharma press release).
Many systemic and topical AD therapies also include hand-specific or hand-adjacent data in their development programs, which helps clinicians make more informed choices when the hands are the primary problem.
For children, the therapeutic landscape has also expanded. Several systemic therapies and multiple topical agents have been studied and approved for younger age groups in recent years, offering more flexibility to tailor regimens for pediatric patients, including those with moderate-to-severe disease or significant impact on quality of life (Source: Incyte; Regeneron; Pfizer press releases).
Practical considerations: safety, monitoring, and patient preferences
Choosing a targeted agent requires balancing expected benefits with safety monitoring needs and patient comfort with potential side effects. For example, JAK inhibitors may require different baseline screening and periodic labs compared with biologic monoclonal antibodies, and patients should be counseled about these practical differences.
Topical nonsteroidal agents can be attractive for long-term local control when safety and convenience are priorities, while systemic agents remain important for extensive or refractory disease. All of these choices should be made in the context of open dialog about goals, risks, monitoring, and cost or access issues.
Looking ahead: genetics, biomarkers, and smarter trials
Emerging tools—such as genetic tests and molecular biomarkers—aim to refine treatment selection between biologics and oral JAK inhibitors, but their routine clinical utility is still under discussion.
Clinicians are hopeful that as biomarkers are validated and incorporated into trials, they will help predict who will respond best to a given therapy, reduce time spent on ineffective treatments, and further personalize care.
Meanwhile, the field is pushing for clinical trials designed to reflect the complexity of typical patients—including comorbidities and real-world treatment sequencing—so that evidence better matches everyday practice.
Bottom line
The current era of AD care is marked by precision and personalization: clinicians now have a richer toolbox that includes targeted biologics, oral JAK inhibitors, and novel topical nonsteroidal agents, enabling more tailored treatment plans aligned with patient needs and safety considerations.
Shared decision-making, awareness of systemic inflammation and comorbidities, and thoughtful selection and sequencing of therapies are key to getting the best outcomes for people living with atopic dermatitis.
Sources
- American Academy of Dermatology Association, Atopic Dermatitis Guidelines (guideline documents and clinical resources).
- Regeneron Pharmaceuticals and Sanofi press materials on dupilumab (FDA approval and clinical program information).
- Pfizer press release and prescribing information for abrocitinib (CIBINQO).
- AbbVie press release and product information for upadacitinib (RINVOQ) in atopic dermatitis.
- Incyte Corporation press releases and FDA communications regarding topical ruxolitinib (Opzelura).
- LEO Pharma press releases related to tralokinumab and topical delgocitinib developments and approvals.
- Eli Lilly press releases and regulatory communications on lebrikizumab.
- Company press materials and clinical trial summaries for topical agents such as tapinarof and roflumilast (Dermavant/Arcutis and other developer resources).
